One of the most critical biological processes is protection and maintenance of the genome. Human chromosomes are constantly exposed to a number of insults and challenges, originating from both intra- and extra-cellular sources. Rapid and effective cellular responses to these diverse genotoxic stresses are essential to ensure genomic stability.
Mammalian cells have evolved multiple protective mechanisms, or DNA repair pathways, to deal with the different types of DNA damage. Each pathway is rapidly activated in response to specific kinds of DNA damage. This highly specific response often involves enzymatic reactions, such as phosphorylation and ubiquitination.
Failure in these DNA repair pathways is detrimental and underlies severe genetic diseases and syndromes in humans. For example, interference with a DNA double strand break (DSB) repair pathway is associated with Nijmegen breakage syndrome, Werner's syndrome and familial breast cancer susceptibility, whereas abrogation of DNA Trans Lesion Synthesis by DNA polymerases is directly linked to the skin cancer syndrome Xeroderma Pigmentosum variant.
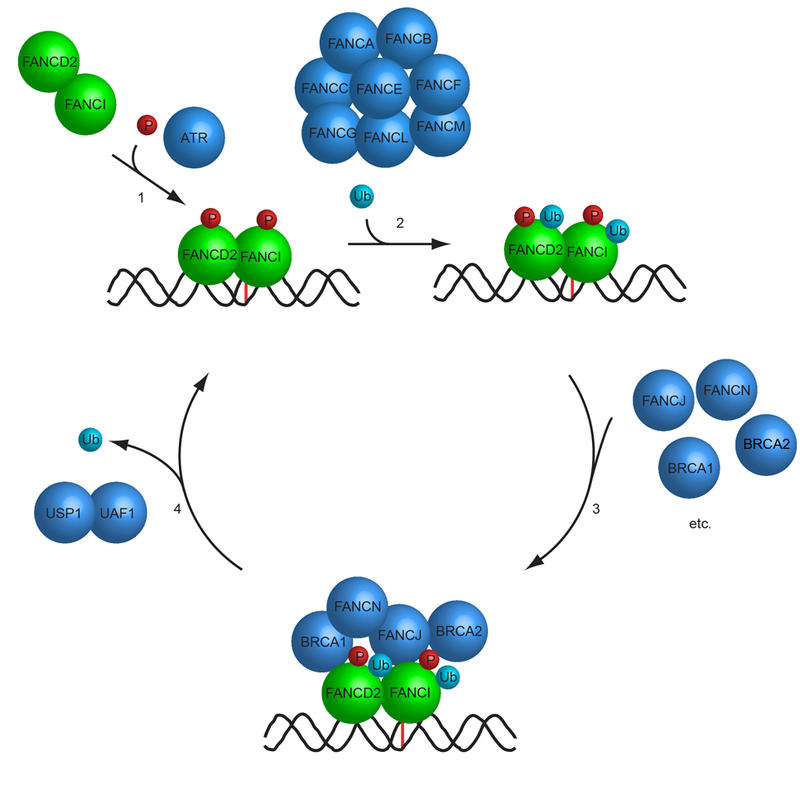
Our laboratory is interested in understanding how the various DNA repair pathways function in human cells. Our previous work has led to the discovery of novel proteins participating in DNA repair pathways such as the Fanconi Anemia DNA repair pathway. Interestingly, our findings demonstrate that these proteins often function as part of large multisubunit protein complexes. Using a combination of molecular and cell biology, live-cell imaging, biochemistry, CRISPR/Cas9 genome engineering, and cryo-EM, we are continuing to identify and study new proteins playing key roles in human DNA repair pathways.
Examples from our research projects:
Figure 1: A) Silver stain of glycerol gradient fractionation of native USP1/UAF1, USP12/UAF1 and USP46/UAF1 deubiquitinating enzyme complexes purified from human HeLa cells. B) In vitro assay measuring the deubiquitinating enzyme activity of the fractions in (A).
Figure 2: Immunofluoresence staining of various DNA repair proteins in human cells before and after DNA damage. Observe how the proteins translocate to damaged chromatin and form the characteristic nuclear foci upon DNA damage.
Figure 3: Live-cell imaging. EGFP-FANCD2 is recruited to DNA interstrand crosslinks (ICLs). Scale bar: 20 μm.
Figure 4: Cryo-EM structure of human FANCD2/FANCI complex showing the Tower domain of FANCD2 and the main body.
Figure 5: Cryo-EM structure of the human FANCD2/FANCI complex